Axon to dendrite4/25/2023 
Results Sodium Influx and Reversal of Sodium Calcium Exchangers Induce Retrograde Spine Loss The extent to which both dendritic spine loss and hyper-excitability use the same retrograde signaling mechanisms is unclear. Axotomy within these platforms produced a delayed enhancement in neuronal hyper-excitability, 1 day following dendritic spine loss. Axotomized neurons within microfluidic chambers also showed that dendritic spine loss ( Gao et al., 2011 Ghosh et al., 2012 Nagendran et al., 2017) and hyper-excitability ( Frost et al., 2015 Nagendran et al., 2017) occur, providing an experimentally tractable model to study initiation and progression of these neuronal injury responses. 
Evidence of ER changes within the soma, called chromatolysis, occurs in axotomized neurons in vitro ( McIlwain and Hoke, 2005 Nagendran et al., 2017). Axotomy performed within compartmentalized platforms produced several characteristic injury responses well described in vivo, including rapid expression of the immediate early gene c-fos ( Taylor et al., 2005) and reduced expression of netrin-1 one day following axon damage. Because of this barrier region, axons can be injured precisely without mechanically disrupting somatodendritic regions and soluble microenvironments can be established for experimental purposes. These devices guide axonal growth of pyramidal cells, through a microgroove-embedded barrier region of almost 1 mm into an isolated axonal compartment. Hippocampal cultures grown within a compartmentalized microfluidic platform provide an injury model system to investigate intrinsic neuronal injury response. 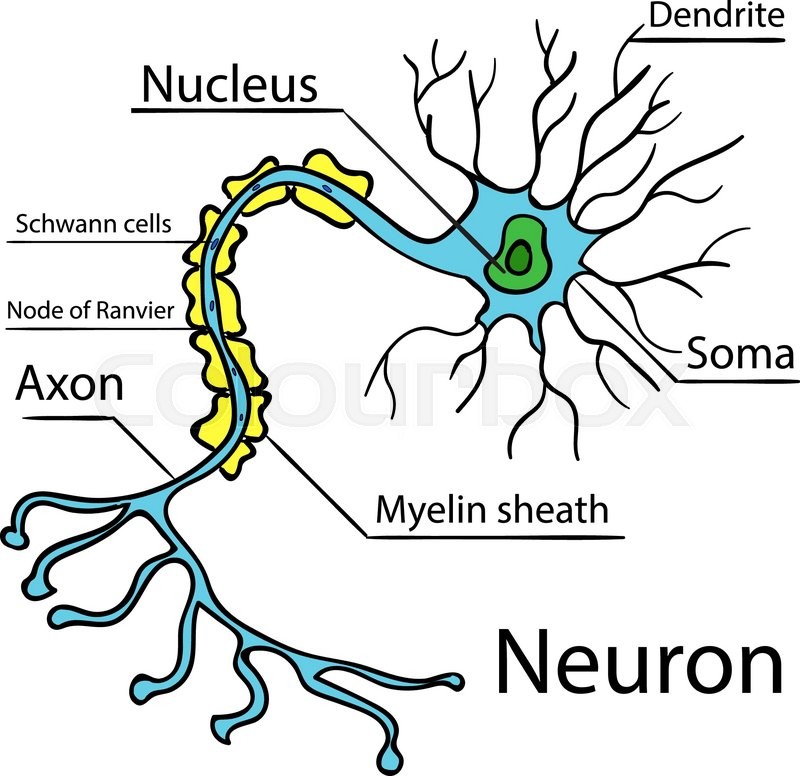
Whether local sodium influx and reversal of NCX during axon damage are needed to transmit signals to the soma to cause dendritic spine loss and hyper-excitability remains unknown. Axon damage of mouse peripheral sensory neurons was reduced with blockade of both sodium channels and the reverse mode of NCX ( Persson et al., 2013), supporting the critical role of NCXs in retrograde injury signaling. Calcium release from smooth ER within the axon may also potentiate axon-to-soma signaling ( Cho et al., 2013 Sun et al., 2014). The increase in sodium ions through voltage-gated sodium channels causes reversal of sodium-calcium exchangers (NCXs) located on the plasma membrane, mitochondria and ER, thus enhancing local intra-axonal calcium levels ( Persson et al., 2013). Breach of the axonal membrane following axon injury causes an influx of ions, including calcium and sodium, into the intra-axonal space. Yet, the intrinsic neuronal signaling mechanisms mediating retrograde synapse loss and hyper-excitability, in particular, remain poorly understood.Īxon injury induces differential gene expression and transcription within the soma, requiring long range signaling from the site of injury to the nucleus ( Rishal and Fainzilber, 2014 Nagendran et al., 2017). These downstream responses are critical for recovery following injury. Axon damage triggers an intra-cellular signaling cascade to effect neuronal injury responses, including axon regeneration, retrograde synapse loss, and hyper-excitability. Further, axonal ER may play a critical and underappreciated role in mediating retrograde hyper-excitability within the CNS.Īcute neural injuries (e.g., stroke, traumatic brain injury, and spinal cord injury) cause profound axon damage. These data suggest that synapse loss and hyper-excitability are uncoupled responses following axon injury. We found that calcium release from axonal ER is critical for the induction of hyper-excitability and inhibition loss. In contrast, sodium influx and NCX reversal alone are insufficient to cause retrograde hyper-excitability. Our data show that sodium influx and reversal of sodium calcium exchangers (NCXs) at the site of axotomy, mediate dendritic spine loss following axotomy. 
To investigate intrinsic injury signaling within neurons, we used an in vitro microfluidic platform that models dendritic spine loss and delayed hyper-excitability following remote axon injury. While axon regeneration is studied extensively, less is known about signaling mediating retrograde synapse loss and hyper-excitability, especially in long projection pyramidal neurons.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
